PDA accounts for 5 to 10% of congenital heart anomalies; the male: female ratio is 1:3. PDA is very common among premature infants (in 45% with birth weight < 1750 g; in about 80% with birth weight < 1200 g). A significant PDA causes heart failure (HF) in 15% of premature infants with birth weight < 1750 g and in 40 to 50% of infants with birth weight < 1500 g.
The Foramen Ovale in Fetal Circulation
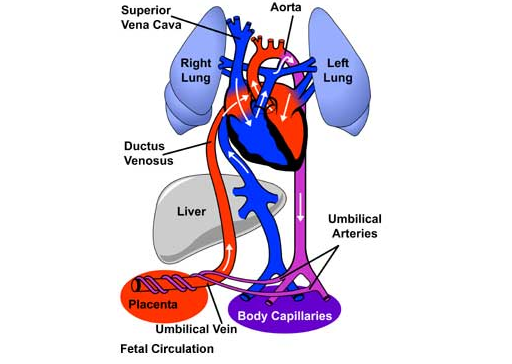
Before birth, the fetal circulatory system includes three open structures through which blood moves that normally close soon after birth. These structures are the ductus arteriosus, the ductus venosus, and the foramen ovale. The foramen ovale allows the shunting of blood from the right atrium into the left atrium.
In the fetus, oxygenated blood (red in the illustrations) enters the circulatory system through placental transfer. The umbilical vein brings oxygenated blood through the ductus venosus to the inferior vena cava and right atrium, where it is directed by the Eustachian valve across the patent foramen ovale and into the left atrium. This allows the left ventricle to pump the most oxygenated blood to the coronary and carotid arteries.
Deoxygenated blood (blue in the illustrations) returning via the superior vena cava is preferentially directed across the tricuspid valve and into the right ventricle. The right ventricle pumps this blood into the pulmonary arteries, and across the patent ductus arteriosus into the descending aorta.
The foramen ovale is an opening in the atrial septum, formed by a flap of septum primum that is pushed open by flow of blood from the right to left atria. Oxygenated blood from the ductus venosus is directed by the Eustachian valve toward the foramen ovale, and into the left heart. This shunts the blood with the highest oxygen content to the left heart for perfusion of the coronary arteries and brain. From the left atrium, the oxygenated blood is pumped into the left ventricle and into the aorta, which carries it to the body. From there it returns to the placenta via the umbilical arteries.
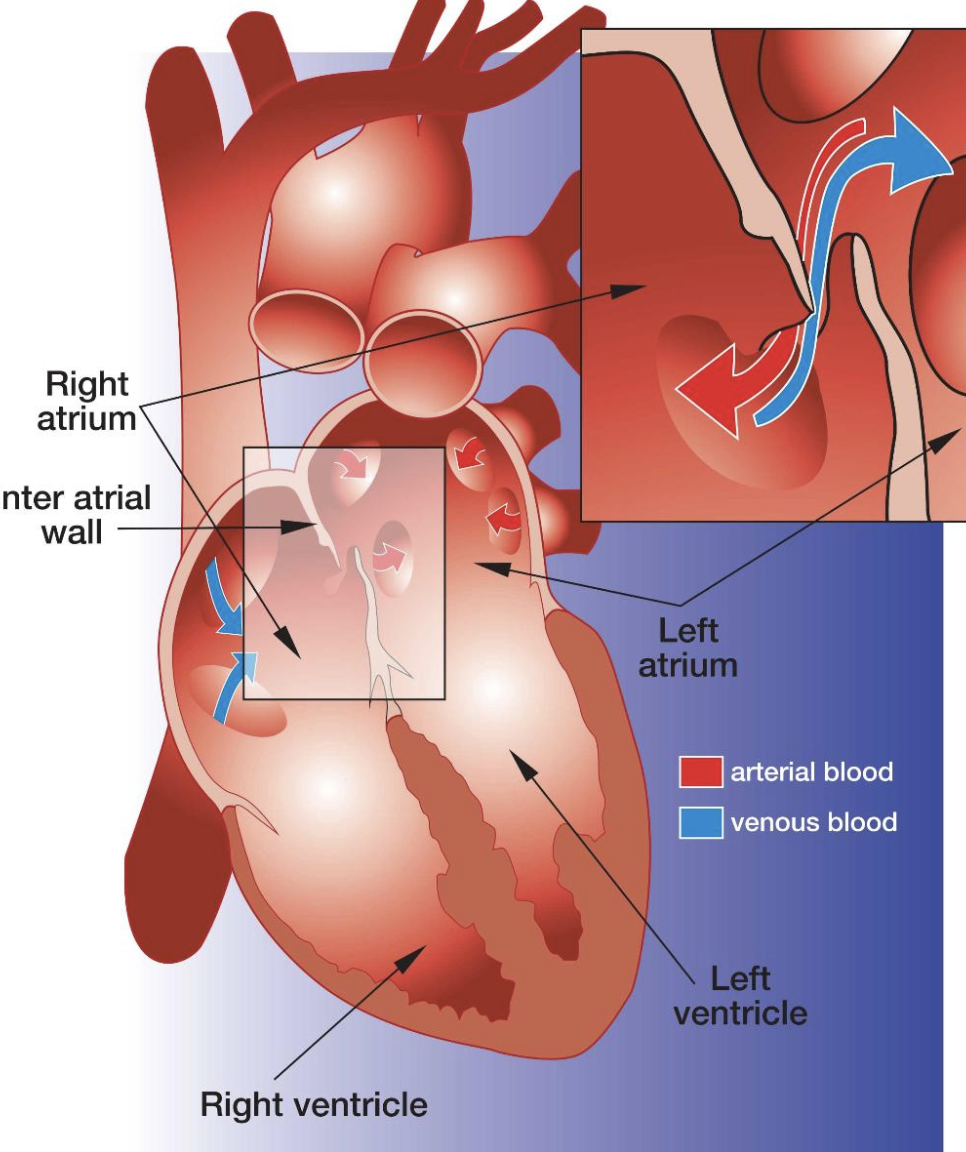
Postnatal Changes
At birth, the lungs inflate, decreasing pulmonary vascular resistance to flow of blood from the right ventricle. The resultant increase in pulmonary blood flow leads to an elevation in left atrial pressure, which normally causes the septum primum flap to seal against the foramen ovale, closing the aperture and effectively separating the two atria. This also increases blood flow to the lungs, as blood entering the right atrium can no longer bypass the right ventricle and is now pumped through the pulmonary artery into the lungs.
Patent Foramen Ovale
Sometimes the septum primum flap fails to seal the foramen ovale after birth and the shunt remains open. This is termed a patent (or open) foramen ovale (PFO). As the heart grows in size, the PFO also increases in size.
The PFO opening allows the shunting of blood between the left and right atria. Generally, because of the higher pressure in the left atrium, oxygenated blood shunts from the left atrium into the right atrium (a left to right shunt). This oxygen-rich blood is then pumped into the right ventricle and through the pulmonary artery to the lungs rather than to the body tissues, which decreases circulatory efficiency.
As many as 27% of all adults have a patent foramen ovale (based on autopsy studies), though only 10-15% are detectable through echocardiography. In most cases, there are no associated symptoms and no treatment is prescribed. However, there is increasing evidence that PFOs are implicated in strokes through embolisation of a thrombus elsewhere. Migraine headaches are also associated with a PFO in some patients.
Treatment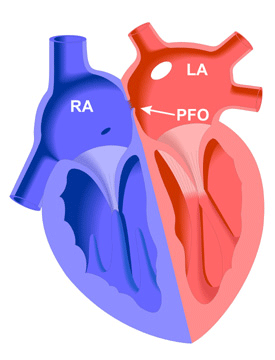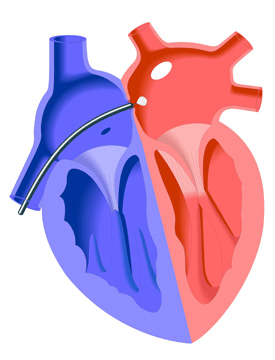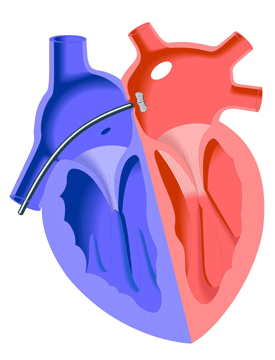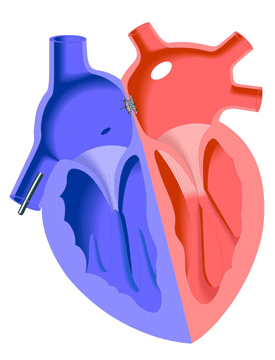
Because of the increased risk of stroke in persons with a PFO, treatment with anticoagulant medications such as aspirin and warfarin may be prescribed. However, there is evidence that the embolism occurs through the opening itself. Therefore, direct closure of the PFO is being increasingly performed in selected patients – especially those with a history of recurrent unexplained strokes due to presumed paradoxical embolism and where pharmaceutical treatment has been ineffective.
PFO closure may be achieved through open heart surgery or through a transcatheter procedure using an occlusion device. The surgical approach involves the closing of the PFO with a double-continuous suture. This procedure is expensive and invasive and often painful. Therefore, the transcatheter approach is used whenever possible.
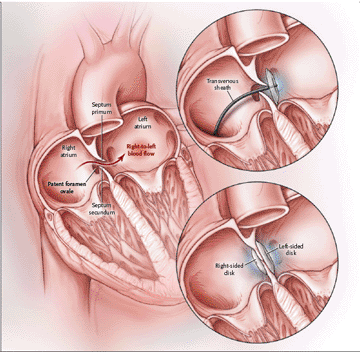
The transcatheter PFO occlusion procedure normally takes from 2 to 4 hours and is performed in a catheterization laboratory. The catheter is inserted in the femoral vein in the groin area, which is numbed with a local anesthetic. Sometimes, general anesthesia or sedation by IV is prescribed.
It is very rare for the body to react negatively to the occlusion device. A few days after the procedure, the patient’s own body tissues will begin to envelope the closure device, a process which is usually complete within 6 months. Even though the PFO is closed by the device, patients are maintained on warfarin and/or aspirin because of the risk of recurrent stroke.
(From: the Cove Point/Johns Hopkins Children’s Center Patient Education for Congenital Heart Disease Site)

Comments 2
Pingback: Cardiac Defects with a Left to Right Shunt (Acyanotic) - Cardiac Health
Pingback: Ventricular Septal Defect (VSD) - Cardiac Health