Tetralogy of Fallot, described in 1888, is seen in about 8% of all congenital cardiac abnormalities. It includes the following defects:
- Pulmonary stenosis (PS)
- Ventricle septum defect (VSD)
- Overriding aorta
- Right ventricular hypertrophy
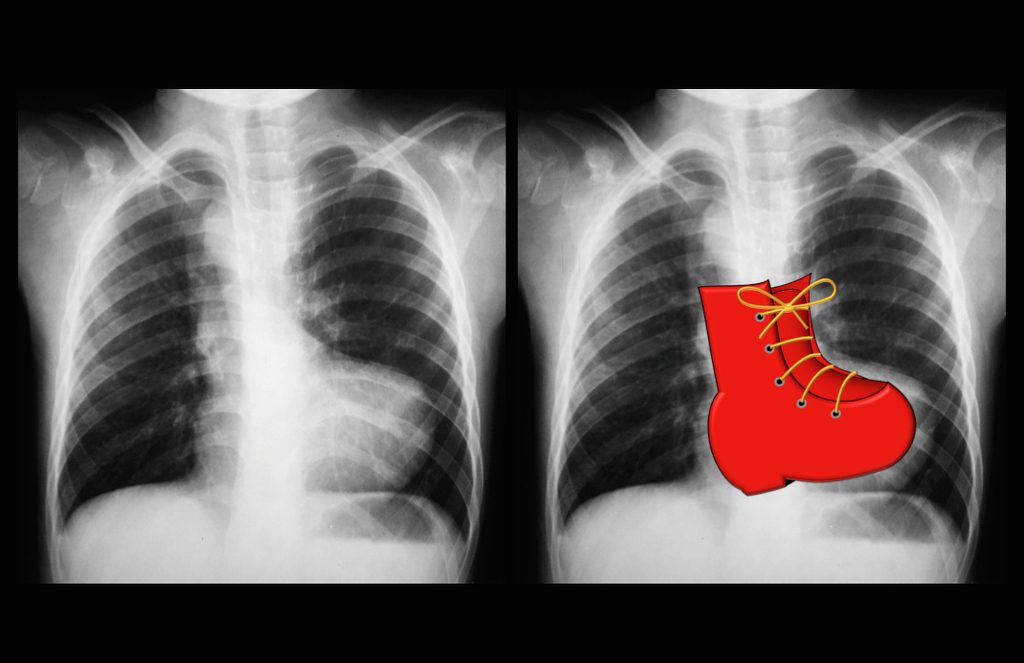
“Boot Shaped heart”. Case courtesy of Dr Vincent Tatco, Radiopaedia.org. From the case rID: 43059
Tetralogy of Fallot Embryology
It is sometimes hard to remember the 4 components of TOF. However, understanding the little bit of embryology that is required to malfunction can result in an easier understanding of why all the four abnormalities in TOF exist.
Remember: the aorta and pulmonary artery start as a single tube the truncus arteriosus which is then divided by the spiral septum. The spiral septum grows down to and attaches to the ventricular septum which isolates the left and right ventricles as well as isolating the aorta and pulmonary artery. However, if the spiral septum is not in the midline, but rather shifted over towards the right side of the heart, then what would happen?
- The opening to the aorta would be large
- The opening to the pulmonary would be small
- The spiral septum would “miss” the ventricular septum
- Poof! One abnormality results in 3 of the “defects” in TOF: “overriding” aorta (it opens up to both the right and left ventricles)
- pulmonary stenosis (the pulmonary artery is small and narrowed)
- VSD (because the spiral and ventricular septa do not line up and connect)
What about the 4th abnormality in TOF: right ventricular hypertrophy? Well, it is a
compensatory result of having pulmonary stenosis. The RV has to work extra hard to
pump blood into the pulmonary artery and therefore hypertrophies to accommodate the
extra work.
The pulmonary stenosis (PS) determines the degree of severity of this defect. Pulmonary perfusion depends on the size of the PS.
In some infants and children, there can be profound narrowing of the right ventricular outflow tract. Because of this it is easier for the blood to cross the VSD right-to-left and go out to the aorta instead of the pulmonary artery. If this is the case, the infant or child can become quite cyanotic.
The most severe form of Tetralogy of Fallot is associated with pulmonary atresia where no blood can cross from the right ventricle to the pulmonary arteries and lungs. In these infants it is necessary to start a medication (Prostaglandin E1) to help keep open the Ductus Arteriosis (PDA) to maintain some bloodflow to the lungs.
Babies with Tetralogy of Fallot may experience intermittent spells of extreme cyanosis, termed “tetralogy spells,” or hypercyanotic spells. These can be serious and even life-threatening.
To increase blood flow to the lungs, an operation known as a Moditfied Blalock-Taussig (B-T) Shunt Procedure can be performed in which a “shunt”, or tiny tube made of Gore-Tex® (yellow in the diagrams to left), is attached between the aorta (or one of its branches) and the pulmonary artery (or one of its two branches – the left pulmonary artery (LPA) or right Pulmonary Artery (RPA):
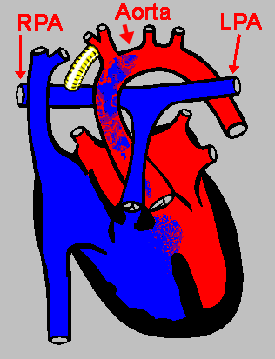
If a shunt procedure was previously performed, the shunt is removed and circulation proceeds more or less as in a normal heart.
Tetralogy patients after surgical repair usually have a murmur of pulmonary insufficiency (diastolic murmur) and residual pulmonary stenosis (systolic). There may also be a murmur of a residual VSD.
Patients after primary surgical repair of Tetralogy in childhood will often require further surgical repairs, especially replacement of the pulmonary valve to minimize right ventricular volume overload as a result of the complete pulmonary insufficiency that is a consequence of the Tetralogy repair.
Patients who have undergone the surgical repair of Tetralogy of Fallot are prone to developing arrhythmias later in life, with more than a third of adult patients experiencing atrial arrhythmias